 
This is because it will increase with decreasing atomic radius, since a smaller atom will have a greater pull on electrons from the positively charged nucleus. Shells 5 through 7: 32 electrons in any known element, however there are additional orbitals available to hold even more electrons, but there is no element with a large enough atomic number to fill up those slots. 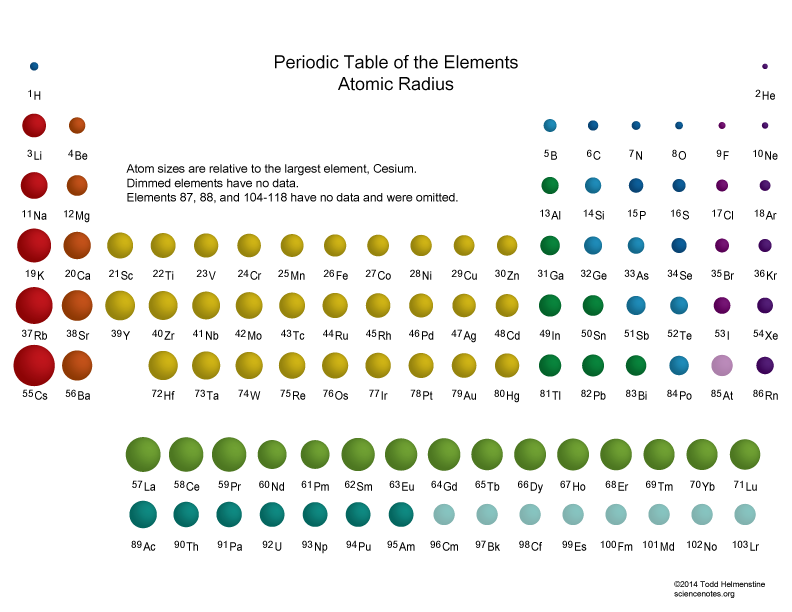
This pattern is the same as ionization energy, increasing as you move up and to the right on the periodic table. For the same reason, ionization energy will also increase moving left to right across a period, since atomic radius also decreases in this pattern.Įlectronegativity: Electronegativity is the ability of an element to attract electrons. This will thus increase moving up on the periodic table, since it will require more energy to take an electron from an inner shell that is closer to the positively charged nucleus. Ionization Energy: Ionization energy is the energy needed to remove an electron from an atom. First Atomic Radius Trend: (Decrease From Left to Right Across a Period) The first periodic trend in atomic radius is a reduction in atomic size as one moves from left to right throughout a period. The atomic radius of atoms generally increases from top to bottom within a group. This is because you are adding more electrons into the same shell, so there is more negative charge which has an increasingly stronger attraction to the positively charged nucleus, pulling the electron shells in closer and thus decreasing the overall atom size. The Periodic Table of the Elements (including Atomic Radius). 
Going across the groups, however, the atomic radius decreases from left to right. Atomic Radius As we move down a group in the periodic table, the number of electron shells increases which makes the atom radii larger. Atomic Radius: Going down the periodic table, each period has higher atomic radius because each period represents an additional electron shell being added, which means the atoms must grow larger. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
